Typically as people start to learn more and more about engines and generator packages, we find them asking: What is a stoichiometric engine, or What is stoichiometry? At the most fundamental level, stoichiometry is a ratio of whole integers. This article will help explain how stoichiometry influences our generator engines.
The Basics of Stoichiometry
Before we begin to talk about engines and generator packages, it helps to explain basic stoichiometry. By definition, stoichiometry is the quantitative relationship (i.e. measurable connection) between a reactant and a product in a chemical reaction. In chemistry, this is a general way of saying what substances are required to fulfill a reaction. A simple example of stoichiometry is:
2 1/2 cups flour + ¾ tsp baking soda + ½ tsp salt, + 2/3 cup sugar + 1 ½ tsp vanilla, + 1 egg, + 1 bag of chocolate chips = The perfect chocolate chip cookie recipe.
For this recipe, or reaction, certain elements are needed to fulfill it. Like in the chocolate chip cookie example, all reactions inside engines are dependent on what they have to start with. In some instances the amount of a compound is unknown, which is where stoichiometry is useful.
This relationship not only determines what is needed for a reaction, but also how it was started. Simply stated, we can look at this like making a grilled cheese sandwich, or in more difficult terms, the combustion of propane.
Let’s continue with another cooking example and say that you are making one grilled cheese sandwich requiring two slices of bread and one slice of cheese. The ratio of bread:cheese is 2:1. Likewise, if you have ten slices of bread, and five slices of cheese, how many sandwiches can you make? We would know the answer is five because we are paying attention to the bread, since the bread:cheese ratio is akin to the air:fuel ratio in an internal combustion engine.
The same concept applies to the combustion of propane (or any hydrocarbon); a certain amount of propane combined with oxygen as a reactant creates the final “product,” which is power generation. An example of how this works is in the following illustration:
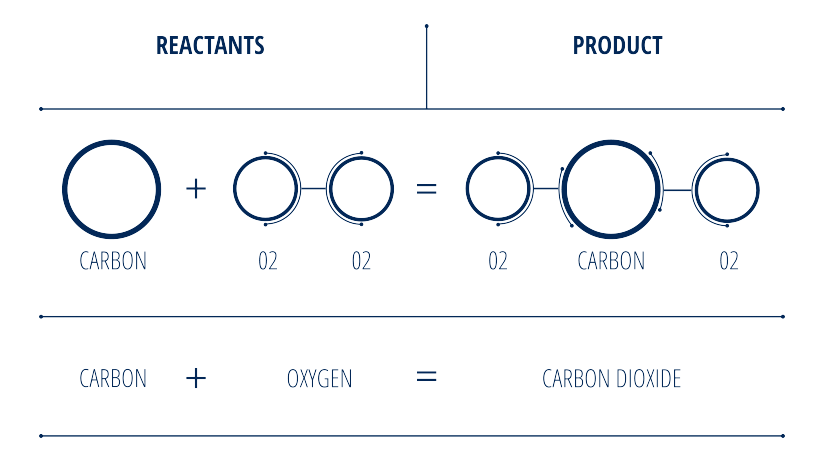
Stoichiometry In A Combustion Engine
Gasoline engines, diesel engines, and natural gas engines can all be stoichiometric. When supplied as a fuel source, the different hydrocarbons (methane, ethane, propane, butane, carbon dioxide, oxygen, nitrogen, and hydrogen sulfide) will cause combustion to vary. Like using the incorrect ratio of ingredients in a recipe results in an inedible cookie, not having the proper ratio of oxygen to fuel results in inefficiency and unused fuel. Unused fuel will be flared which results in higher emissions.
Knowing the type of hydrocarbons used allows for a simple calculation, based on the properties of said hydrocarbons. However, reactions that do not have correct/optimal ratios of fuel and oxygen would not technically be considered stoichiometric.
The Consistency of Stoichiometry
Mesa natural gas generators are designed to create a stoichiometric environment by constantly adjusting the ratios of fuel to air based on the quality of the gas that they receive, and our products do this in real-time. This is what makes our units superior and reliable.
A true stoichiometric engine is ideal because of the consistency it provides. As opposed to fluctuating in real time, a day-to-day fluctuation of burning specifically rich or lean could lead to inefficiency, engine damage, and higher NO2 oxides that are detrimental to health, atmospheric conditions, and plant life. Stoichiometry varies between different hydrocarbons.
For instance, propane has a ratio of 15:1 whereas ethanol has a ratio of 9:1. This is to say in the example of propane, 15 parts propane and 1 part air make up the stoichiometric ratio for that hydrocarbon. No two wellheads are the same, which is why Mesa natural gas generators have the ability to calculate stoichiometric mixture in real time; this is a key factor in efficiency, longevity, and reliability.











